There Is No Single Cause of Autism - There Is a Shared Biochemical Cascade
Each individual has their own combination of genetic and epigenetic factors that contribute to stress-response activation.
Individual Inputs
Autism does not come from one cause. It emerges from a unique combination of genetic and epigenetic factors in each individual.
Shared Mechanism
These factors converge on the stress-response system, which is biologically conserved across humans and across species.
Consistent Biology
Because the stress-response system is conserved, the way it impacts the body follows consistent biological patterns.
Predictable Outcomes
While each individual’s inputs are different, the downstream effects of sustained activation are predictable based on those variables.
The model in one sequence
The framework proposes a structured sequence linking genetic/epigenetic regulatory system domain activation, biochemical pathway shifts, temporal system domain disruption, and the clustering of autism traits and comorbid conditions.
Autism traits and comorbid conditions do not occur randomly. They cluster in consistent patterns across individuals.
These patterns emerge from shifts in biochemical pathway activity under stress, altering how physiological systems allocate resources.
The model organizes these shifts into a cascade centered on BH4-dependent pathways, regulatory system domain activation, and its impact on temporal system domains.
If the structure is correct, independent research should converge on the same mechanisms, pathways, and system interactions.
How This Theoretical Model Was Built
Kitzerow’s Autism and the Comorbidities Theoretical Model was developed through the Jigsaw Puzzle Research Methodology, a computational systems analysis approach that builds a conserved species-level biochemical reference framework first, then compares demographic-level biomarker findings against that framework to identify recurring points of dysregulation and reconstruct a coherent biochemical cascade.
Rather than treating biomarkers, pathways, and studies as isolated findings, this methodology treats them as pieces of a larger biological structure that must be assembled into a coherent functional model.
A biochemical network of gene-coded proteins is constructed to define the conserved species-level functional blueprint used as the reference system.
Demographic-level biomarker datasets are mapped onto that reference framework so population-level variation can be compared against shared biological architecture.
Recurrent deviations from the conserved framework are identified as consistent points of dysregulation across datasets, pathways, and regulatory systems.
Those recurring patterns are traced across pathways to reconstruct the biochemical cascade linking autism traits and comorbidities to shared system-level dysregulation.
Kitzerow’s Autism and the Comorbidities Theoretical Model
Autism and its comorbidities emerge from a shared biochemical cascade in which different genetic and epigenetic inputs converge on the same regulatory system shift.
Regulatory System Activation
Individual genetic and epigenetic inputs vary. What remains consistent is activation of regulatory systems involved in survival, adaptation, and stress response.
The BH4 Shunt
Because GCH1, the rate-limiting step of BH4 synthesis, is redox sensitive, regulatory system activation alters BH4 availability. This drives the BH4 Shunt and redirects biochemical resources toward survival over typical development and typical function.
Autism Traits and Comorbid Traits
Once BH4 is redirected, downstream shifts emerge across multiple systems. Autism traits and comorbid traits cluster because they are outputs of the same constrained biochemical state.
What Remains Consistent Across Individuals
The inputs are not identical from person to person. The shared pattern is regulatory system activation and the BH4 Shunt, with biochemical resources being prioritized toward survival rather than typical development and typical function.
How the Cascade Unfolds
The model is structured as a regulatory cascade in which system activation drives a BH4-dependent shift that organizes downstream pathway behavior and outcomes.
What Determines Cascade Behavior
The cascade is shaped by how systems are activated, how long they remain active, and how that activation interacts with biological timing.
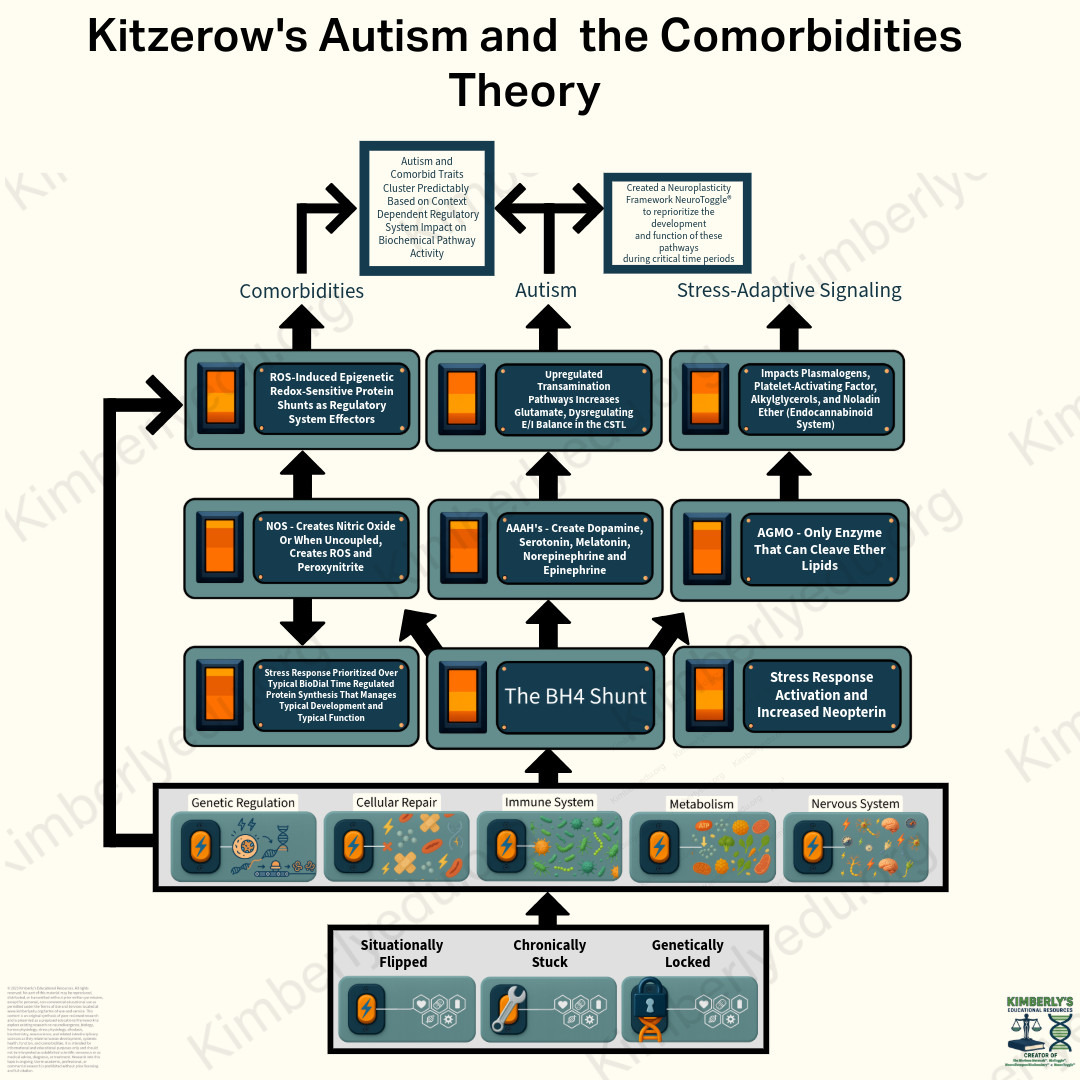
Kitzerow’s Autism and the Comorbidities Cascade
This cascade shows how regulatory system activation can shift biological resource allocation through the BH4 Shunt, prioritize survival effectors, disrupt BioDial activity, and alter function and development across regulatory system domains within a multivariable stress-response system cascade.
Regulatory system activation can begin or persist in different ways
These patterns describe whether activation is built into the system, fails to resolve, or resolves after a temporary trigger.
Activation occurs across regulatory system domains
These regulatory system domains are BioToggles. They are the core domains involved in survival, function, repair, regulation, and development.
The regulatory loop detects stress and initiates a response
The body detects a change, compares it against a setpoint, interprets the signal, and activates effectors to restore regulation.
The BH4 Shunt reallocates biological resources
The BH4 Shunt acts as a central resource allocation mechanism. Under activation, BH4-dependent pathways shift toward survival-focused regulatory effectors.
Regulatory domains shift toward survival-focused effectors
Resources are routed toward effectors needed for stress resolution. Survival responses are prioritized over typical development, repair, maintenance, and day-to-day function.
BioDial activity is disrupted
BioDials represent the ongoing flow of protein synthesis. When BioToggles are activated, timing-based synthesis patterns are deprioritized as regulatory effectors are prioritized.
Gene expression shifts toward survival pathways
Gene expression adapts to support survival pathways and conserve resources, changing which proteins and pathways are prioritized over time.
Each regulatory domain is impacted through function and development
Altered BioToggle activity and disrupted BioDial activity work together to change both how each regulatory domain works and how it develops, repairs, and adapts over time.
Trait outcomes emerge
Altered nervous system development → core autism traits
When regulatory activation alters nervous system function and development during development, the outcomes appear as core autism traits.
Other regulatory domains → comorbid traits
When regulatory activation alters immune, metabolic, cellular repair, genetic regulation, or other regulatory domain activity, the outcomes appear as comorbid traits.
Autism traits and comorbid traits reflect how a multivariable stress-response system cascade is expressed across regulatory system domains and biological timing systems.
Autism and the Comorbidities Theory
The outcomes of the model
Autism aligns with the genetic domain, while neurodivergent traits and comorbidities emerge across other domains based on activation patterns.
How Comorbid Traits Are Grouped in This Model
Comorbid traits are not random or isolated. They tend to cluster based on the biological systems they affect.
In this model, comorbidities are organized by regulatory system domains. This makes it easier to see how different symptoms connect within the same underlying biological patterns.
The categories below show how comorbid traits group together across systems rather than appearing independently.
What Are the BioToggles?
Kitzerow categorized the regulatory system domains into five categories that she named the BioToggles. The comorbidity categories below are organized within those five domains.
Immune System Differences Can Show Up Alongside Autism
In this framework, immune-related comorbidities fall under the immune system domain because they affect inflammation, immune signaling, and how the body responds to stress and illness.
What It Regulates
Inflammation, immune response, illness signaling, and how the body reacts to internal and external stressors.
What It Can Look Like
Autoimmune patterns, autoinflammatory responses, frequent illness, strong inflammatory responses, or broader immune dysregulation.
Why It Matters
When this system is persistently activated, it can affect regulation across the body and contribute to broader physiological stress patterns.
Relevant Sources
Meltzer and Walter, 2017
Park et al., 2025
Erbescu et al., 2022
In this model, immune comorbidities are part of the broader biological pattern, not separate add-ons.
Metabolic Differences Can Shape Daily Function
In this framework, GI and metabolic comorbidities fall under the metabolic domain because they affect digestion, nutrient handling, energy production, and physical regulation.
What It Regulates
Digestion, nutrient use, energy production, metabolic balance, and how the body fuels development and function.
What It Can Look Like
GI problems, food sensitivities, unstable energy, feeding issues, metabolic differences, or obesity.
Why It Matters
When metabolism is strained, the effects can show up in both daily function and broader physical regulation.
Relevant Sources
Al-Beltagi, 2021
Khachadourian et al., 2023
Aziz-Zadeh et al., 2025
In this model, GI and metabolic patterns are part of the same systems picture, not separate unrelated problems.
Cellular Repair Differences Affect Structure and Recovery
In this framework, connective tissue and repair-related comorbidities fall under the cellular repair domain because they affect structural integrity, resilience, and how the body maintains itself.
What It Regulates
Connective tissue integrity, structural support, tissue maintenance, and physical repair processes.
What It Can Look Like
Joint instability, slow recovery, chronic pain, tissue fragility, or reduced or altered pain perception.
Why It Matters
Structural and repair differences can shape physical stability, comfort, and how stress is carried through the body.
Relevant Sources
In this model, chronic pain or altered pain perception can be part of the same repair and structural pattern.
Nervous System Differences Affect Regulation and Stability
In this framework, autonomic, psychiatric, and neurological comorbidities fall under the nervous system domain because they affect internal regulation, state stability, and how the body responds to demand.
What It Regulates
Stress response, autonomic function, emotional processing, neurological stability, and internal state regulation.
What It Can Look Like
PoTS, anxiety, OCD, ADHD, seizures, tics, FND, burnout, dysregulation, or unstable nervous system states.
Why It Matters
When nervous system regulation is unstable, the effects can shape cognition, emotion, movement, and daily function all at once.
Relevant Sources
Owens et al., 2021
Khachadourian et al., 2023
Soghomonian, 2024
Owens et al., 2021
In this model, nervous system comorbidities reflect system-level instability, not isolated failures of will or character.
Genetic Regulation Shapes Timing Across the Body
In this framework, sleep and circadian-related comorbidities fall under genetic regulation because they reflect timing, coordination, and regulation across biological systems.
What It Regulates
Sleep timing, circadian rhythm, biological coordination, and broader regulatory timing across development and daily function.
What It Can Look Like
Sleep disorders, irregular energy rhythms, circadian disruption, or timing-related learning and regulation differences.
Why It Matters
Timing systems influence when biological processes happen, so disruption here can ripple across the rest of the body.
Relevant Sources
In this model, circadian and sleep differences are part of broader regulatory timing, not just isolated sleep issues.
What This Model Explains
The value of a model is not just how it is structured, but what it consistently explains.
Why autism traits occur together
Traits cluster because they emerge from the same constrained biochemical cascade rather than independent causes.
Why comorbid conditions co-occur
Comorbidities follow predictable patterns because multiple systems are shifted simultaneously under shared regulatory activation.
Why presentation differs between individuals
Differences in activation source, duration, and timing across systems produce variation in traits and outcomes.
Why development follows different paths
When regulatory systems prioritize survival, neural development is shaped differently, altering how circuits form and function.
Why outcomes can still change
Because neural circuits are plastic, targeted input can still build and refine function even after initial development.
Implications of This Model
When a system is understood at the level of its underlying structure, it changes how outcomes are interpreted and approached.
Traits reflect system state
Observable traits reflect the state of underlying biological systems rather than isolated differences in behavior or ability.
Development follows system priorities
When regulatory systems prioritize survival, development follows a different trajectory based on those conditions.
Differences are structured, not random
Individual differences reflect variations in activation, duration, and timing rather than unpredictable or unrelated factors.
Outcomes depend on what is targeted
Approaches that align with underlying mechanisms can influence outcomes more effectively than those focused only on surface-level behavior.
Patterns can be anticipated
When the system is understood, patterns across traits and comorbidities become predictable rather than disconnected.
How This Model Is Supported
A model gains strength when independent findings align with its structure and predictions.
Pathway-level consistency
Independent research continues to identify disruptions in neurotransmitter synthesis, redox balance, and regulatory system function consistent with this cascade structure.
E/I balance in CSTL circuitry
Studies targeting excitatory and inhibitory balance in CSTL circuits align with the downstream effects predicted by the AAAH Shunt.
Comorbidity clustering
The consistent co-occurrence of metabolic, immune, and neurological conditions reflects coordinated shifts across regulatory systems.
Updated frameworks aligning
Recent updates to existing models have begun to reflect cascade-based structures that mirror this model’s organization of stress activation, pathway shifts, and downstream effects.
Testable Pillars of Kitzerow's Autism and the Comorbidities Theoretical Model
This model can be evaluated at defined system-level stages where regulatory activation, pathway behavior, neural circuitry, and downstream outcomes converge. These pillars are integration points built on mechanisms already studied at the micro level, allowing the cascade to be tested through how those mechanisms interact across systems and over time.
Stress Activation
Genetic and epigenetic factors activate internal stress-response systems across regulatory domains, including the immune system, metabolism, cellular repair, nervous system, and genetic regulation.
These activations may be situational, chronic, or genetically driven. The duration and category determine downstream biological effects.
Do genetic and epigenetic mutations produce a convergent and sustained stress-response state across regulatory systems?
BH4 Pathway Shunt
Stress-response activation redirects biochemical pathway activity through the redox-regulated, GCH1-mediated BH4 Shunt, shifting activity across AAAH, NOS, and AGMO pathways.
This coordinated redistribution links multiple physiological systems and creates shared biochemical conditions underlying both autism traits and comorbid conditions.
Does stress-induced BH4 pathway redirection produce biochemically linked autism and comorbid trait clustering?
Neural Circuit Disruption
The AAAH pathway shifts aromatic amino acids away from monoamine synthesis and toward glutamate production, altering neurotransmitter balance.
This contributes to excitatory and inhibitory imbalance within cortico-striatal-thalamic circuitry, which drives the expression of autism traits.
Does disruption of excitatory and inhibitory balance within CSTL circuitry produce autism traits?
Comorbidity Clustering
NOS Shunt-induced epigenetic redox-sensitive protein shunts function as regulatory effectors that alter biochemical pathway activity across systems.
These shifts disrupt coordination across biological timing cycles and produce consistent clustering of autism traits and comorbid conditions over time.
Do genetic and epigenetic factors alter biochemical pathway activity, producing consistent clustering of autism and comorbid traits?
Where to Start
Choose the path that matches what you are looking for.
Review the Evidence
Full framework, methodology, validation, and supporting data.
Start Here → ParentsSupport Your Child
Understand development, learning, and practical application.
Start Here → EducatorsTransform Instruction
Apply neuroplasticity-based teaching to build skills and behavior.
Start Here → NeurodivergentsNeurodivergent Biochemistry
See how the model explains development, function, and experience.
Start Here →Common Questions About the Model
These questions break down the model step by step, from core concepts to mechanisms, outcomes, and evidence.
What is this theory, simplified?
Autism and comorbid traits arise from the same upstream biochemical shift. That shift alters development and function in predictable ways, producing both autism traits and comorbid patterns.
Why does the model link autism and comorbidities?
Because they co-occur at high rates. That level of co-occurrence suggests a shared underlying mechanism rather than unrelated conditions.
Is there one single cause of autism?
No. Each individual has a unique combination of genetic and epigenetic factors, but those factors converge on the same stress-response system.
Core Mechanism Questions
These questions define the central biochemical regulators used in the model and clarify how BH4 is positioned within broader cross-system pathway control.
What is BH4?
BH4 is a cofactor that acts as a regulatory molecule across critical biological pathways. It supports neurotransmitter synthesis, impacting mental health and autonomic regulation; nitric oxide signaling, influencing immune function and genetic and epigenetic regulation; and ether lipid catabolism, affecting the endocannabinoid system. Together, these pathways link multiple systems through shared biochemical regulation. Because of this, changes in BH4 availability can produce coordinated effects across both neural development and systemic function.
What is the BH4 Shunt?
The BH4 Shunt describes the redirection of BH4-dependent pathway activity under stress, shifting how biological resources are allocated across systems.
What are redox-sensitive protein shunts?
These are regulatory effectors that alter protein function based on cellular conditions, shifting pathway activity across systems.
Pathway Logic Questions
These questions explain why specific BH4-dependent pathways are prioritized in the model and how pathway shifts affect development, regulation, and comorbidity formation.
Why does the theory focus on AAAH, NOS, and AGMO?
These pathways are the primary BH4-dependent pathways and are not utilized in other regulatory contexts. AAAH governs neurotransmitter synthesis, NOS regulates nitric oxide signaling and redox balance, and AGMO controls ether lipid metabolism and endocannabinoid-related processes. Because all three depend on BH4, shifts in BH4 availability simultaneously alter activity across multiple systems, linking neural, immune, and metabolic function within the same mechanism.
How do these BH4-dependent pathway shifts specifically impact development and function?
The BH4 Shunt redirects activity across three BH4-dependent systems, and each one affects a different biological domain.
1. Neurotransmitter system (AAAH): Reduced BH4 availability shifts aromatic amino acid processing away from monoamine synthesis, lowering dopamine, serotonin, and melatonin, while increasing glutamate. This disrupts excitatory and inhibitory balance within cortico-striatal-thalamic-limbic circuits that regulate movement, habits, reward, and behavioral control, producing autism traits.
2. Ether-lipid metabolism (AGMO): Reduced ether-lipid breakdown alters stress-adaptive lipid signaling, including the endocannabinoid system and broader lipid balance. This changes how the body regulates adaptive responses across systems.
3. Nitric oxide and redox system (NOS): Increased oxidative stress activates redox-sensitive protein shifts that reallocate biological resources toward stress adaptation instead of typical development and maintenance. Over time, this produces cumulative strain, allostatic overload, and wear and tear, leading to comorbid conditions.
Because this stress state is driven by genetic and epigenetic factors, it remains active, creating a lifelong allostatic pattern that produces both autism traits and predictable comorbid clustering.
The specific autism and comorbidity phenotype depends on which regulatory stress systems are activated, when activation occurred during development, and how long it persists.
Trait Expression Questions
These questions explain how autism traits and comorbid traits emerge in the model, and why expression varies across individuals.
How do autism traits emerge in this model?
Autism traits emerge from altered neural development driven by shifts in BH4-dependent neurotransmitter pathways.
Reduced monoamine synthesis, including dopamine, serotonin, and melatonin, combined with increased glutamate production, disrupts excitatory and inhibitory balance within cortico-striatal-thalamic-limbic circuits. These circuits regulate movement, habit formation, reward processing, and behavioral control.
When these pathways are altered during critical developmental windows, neural circuit formation is affected. Because skills and behaviors are encoded within these circuits, this results in the observable traits associated with autism.
How do comorbid traits emerge in this model?
Comorbid traits emerge from sustained activation of regulatory system domains under a chronic stress-response state.
BH4-dependent shifts in nitric oxide signaling and redox balance activate redox-sensitive protein effectors, which reallocate biological resources toward stress adaptation rather than typical maintenance and repair. This alters function across systems including immune, metabolic, autonomic, and cellular repair processes.
Over time, persistent activation produces cumulative strain, instability, and wear, referred to as allostatic overload, resulting in comorbid conditions that cluster across regulatory systems.
Why do traits vary between individuals?
Variation occurs because stress-response activation is not uniform across individuals.
Different genetic and epigenetic factors influence which regulatory system domains are activated, which BH4-dependent pathways are most affected, and which redox-sensitive protein effectors are engaged. Activation can also vary in intensity, timing, and duration.
Because development is time-dependent and systems are interconnected, differences in when activation occurs and how long it persists produce distinct patterns of autism traits and comorbid trait clustering in each individual.
Model Development and Validation Questions
These questions explain how the model was built, what is established versus novel, and what level of validation currently supports the framework.
How was this model developed?
This model was constructed using raw biological data derived from existing research. A biochemical network of gene-coded proteins was built to map functional relationships across regulatory systems.
Autism biomarker data was then compared against this network to identify consistent patterns of dysregulation. This process revealed a structured biochemical cascade linking stress-response activation, BH4 pathway redirection, neural circuit disruption, and comorbidity clustering.
This approach identifies system-level structure within existing data rather than generating new experimental datasets.
What was already established in existing research?
Prior research had already established several relevant mechanisms, including oxidative stress, BH4-dependent pathway function, neurotransmitter imbalance, nitric oxide signaling, immune activation, metabolic disruption, and redox-sensitive protein regulation.
These findings were derived from experimental and clinical studies and represent the underlying biological data used in constructing the model.
However, they were studied across separate domains and were not previously organized into a unified framework explaining their interaction or co-occurrence.
What is novel about this model?
The novel contribution of this model lies in its structure, not in the discovery of individual mechanisms.
It organizes previously established biological findings into a unified biochemical cascade defined by four core pillars: stress-response activation, BH4 pathway redirection, neural circuit disruption, and comorbidity clustering.
This framework explains how autism traits and comorbid conditions emerge from the same upstream process through coordinated pathway shifts and system-level prioritization.
What parts of the model have been validated?
The model has received mechanistic validation at the level of its individual components and their relationships.
Beginning in 2025, emerging research has supported the mechanisms corresponding to each of the four pillars. This includes evidence consistent with stress-response activation, BH4 pathway shifts, neural circuit disruption, and system-level effects associated with comorbid conditions.
This validation reflects that the relationships described within the model are consistent with observed biological processes.
What has not yet been fully validated?
The complete system-level model has not yet been validated as a single integrated framework.
While individual mechanisms and their relationships are supported, full validation would require direct testing of the entire biochemical cascade operating together within the same system.
It is important to distinguish between validating individual mechanisms and validating a fully integrated systems-level model. However, in biochemical networks, validation of individual nodes and their interactions provides convergent support for the integrity of the overall system architecture.
How does this model compare to existing biomedical autism models?
Existing biomedical autism models have identified multiple relevant mechanisms, including oxidative stress, mitochondrial dysfunction, immune activation, metabolic disruption, and neurotransmitter imbalance. These findings are well-supported in the literature and are widely recognized as contributing to autism and related conditions.
However, these models typically examine these mechanisms either in isolation or as loosely connected contributors. Even when integrated frameworks are proposed, they generally describe associations between systems rather than defining a specific biochemical cascade that links them in a stepwise and testable structure.
For example, oxidative stress and mitochondrial dysfunction are consistently reported in autism, with evidence showing their interaction with immune signaling and neural development, but the precise organization of these interactions into a unified causal architecture remains unclear.
In contrast, this model was constructed by mapping raw biological data into a biochemical network and identifying how these mechanisms align within a defined sequence. It organizes previously established findings into four core pillars: stress-response activation, BH4 pathway redirection, neural circuit disruption, and comorbidity clustering.
The key distinction is that this framework defines a structured biochemical cascade that links autism traits and comorbid conditions through shared upstream processes, rather than treating them as parallel or loosely connected outcomes.
Where can I read the supporting research?
Supporting research is available through curated validation pages and primary source citations.
These include detailed breakdowns of validated mechanisms as well as direct links to the scientific literature supporting each component of the model.