Independent Replication of My Work
& Originator Erasure
Originator Erasure
I documented my discovery process through a timestamped public living documentary to establish verifiable originator evidence. As has happened throughout scientific history, institutions have begun reproducing core components of my framework without attribution.
We teach this in elementary school: if a source is not credible, it should not be used. If it is credible enough to use, the original source must be cited. That is standard academic practice.
Frequently Asked Questions
Still have questions? Take a look at the FAQ or reach out anytime at kitzerow@kimberlyedu.org
-
My work is not a literature review. It is a bioinformatics analysis built from raw UniProt protein data, where I constructed a biochemical network and derived novel conclusions. This makes it a primary source. I am the first to state these conclusions, which is why those replicating my frameworks leave the text containing my conclusions uncited because there is no one else they can reference.
-
I got involved in autism research after my nonverbal autistic daughter’s nonverbal autism diagnosis. It was 2020 and all in person services were shut down due to covid, so I was the only chance she had. I graduated with degrees in education and special education, and a minor in instruction strategies, from UW Superior (summa cum laude.) I went back to school for bioinformatics for help after I developed my Autism and the Comorbidities Theory in 2023. I earned all As. I was advised not to pursue a PhD at this time because any further work would be owned by my supervising professor. To maintain scientific integrity and retain ownership of my intellectual property, I have to stay independent.
-
The Matilda Effect has been studied and determined as dangerous for a plethora of ethical and scientific reasons.
If you paid off your car, would you be upset if someone stole it? What if someone told you to let them steal it, and just share the car from now on, even though they never paid it off and initially stole it?
I spent thousands of hours of my life on this, bet my reputation on it publicly, and did it in my disabled daughters name. It’s mine because l paid for it with my blood, sweat, and tears. They did not. Im not going to let someone steal my car or share it with people willing to steal something that isn’t theirs .
-
Absolutely! Replication, derivatives, and falsification are cornerstones of the scientific method. . It is how we check for accuracy within a framework or data.
The problem isn’t the fact that it is being replicated, falsified, or turned into derivatives. The problem is that these researchers are claiming they originated the concept and their affiliated colleges are putting out press releases claiming credit for work I did in my disabled daughter’s name.
-
My Discovery
I discovered one unified system for autism phenotypes you can find the full timeline of discovery here, and a full breakdown here:
What Is Novel About My Framework
The Exclusivity Principle
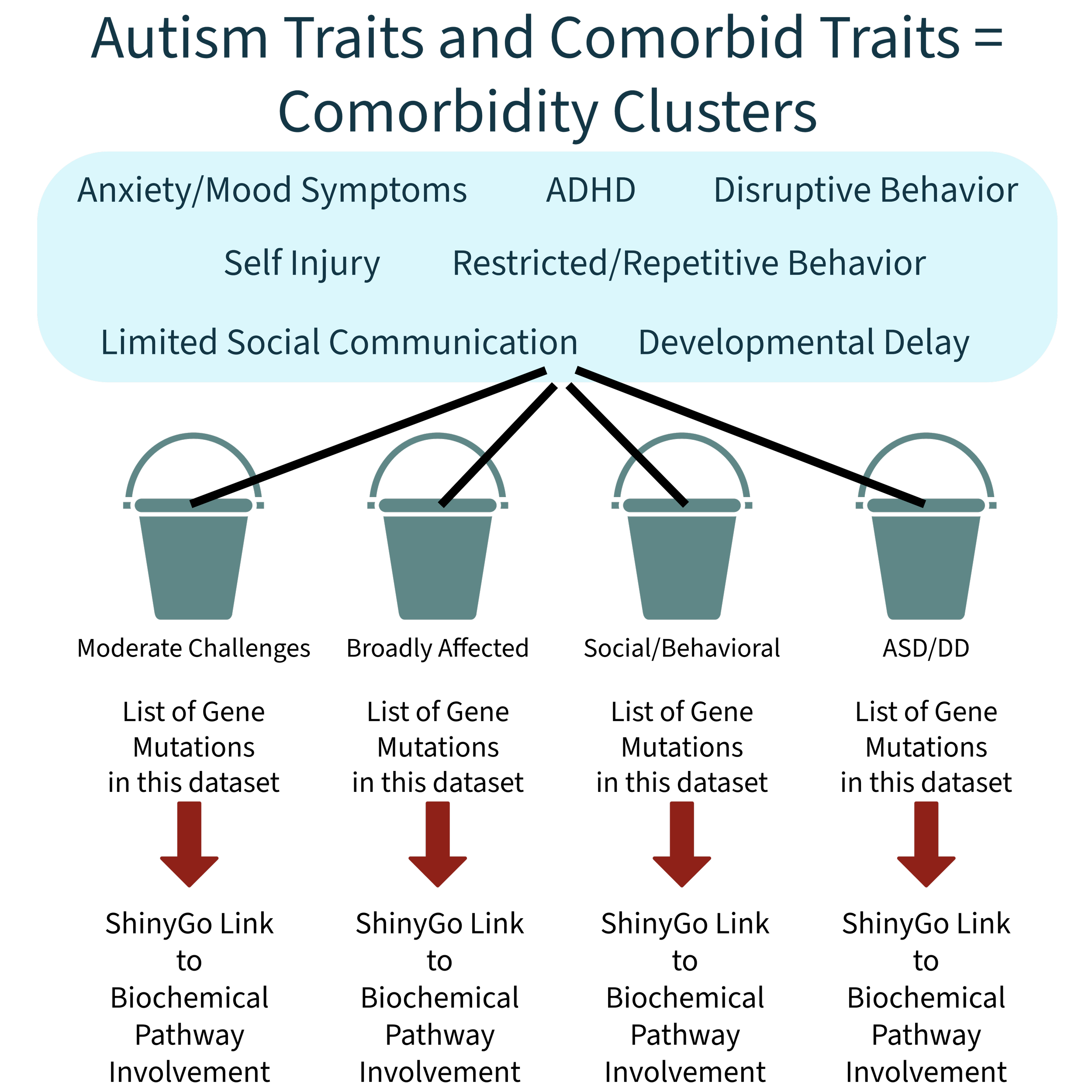
My initial hypothesis is what is now the Exclusivity Principle. It states that autism traits and comorbidities appear together because they arise from the same underlying gene-pathway mechanism. This principle predicts that every autism phenotype will show:
• its own set of gene mutations
• its own epigenetic signature
• its own pattern of pathway activity
• its own predictable cluster of autism traits and comorbiditiesBefore my work, no model proposed this mechanistic inseparability. Autism was described through traits, behaviors, and statistics rather than a unified biological system with predictable trait outcomes. The Exclusivity Principle is the starting point from which my full framework emerges, culminating in what I call The Autism and the Comorbidities Theory.
A Mechanistic Chain Linking Genes to Traits
My framework is the first to connect these all of the levels of molecular biology into a single causal sequence. It traces:
• the gene mutations that define each phenotype
• the epigenetic shifts produced by those mutations
• the resulting pathway behavior shaped by those regulatory settings
• the autism traits and comorbidities that emerge from that biochemical stateThis chain explains why traits consistently cluster, why comorbidities are inherent to autism, and why each phenotype presents with its own pattern.
The Novel Mechanistic Bridge
The central innovation in my work is the bridge between genetics, biochemical pathway activity, and lived phenotype.
No previous model explained how categories of gene mutations alter pathway function that physiologically manifests into specific autism traits, comorbid traits, and phenotype-level patterns. My framework provides that missing mechanistic step.The Logical Structure of My Framework
The downstream flow from hypothesis to conclusion is as follows. You can also find how I applied the scientific method here:
Exclusivity Principle
It is implausible for autism traits and comorbid traits to co-occur without a joint root mechanism in each phenotype, arising from the same biochemical mechanism.Gene Mutations
Each phenotype begins with its own mutation pattern.Regulatory System Breach
These mutations impair one or more regulatory systems within the five BioToggles: immune system, metabolism, cellular repair, nervous system, and genetic regulation.Epigenetic Response
The regulatory breach triggers phenotype-specific epigenetic adjustments.Biochemical Pathway Activity
Epigenetic settings reshape pathway behavior. This includes activation of the BH4 Shunt, which reallocates resources across all five BioToggle systems and alters downstream biochemical function according to the specific regulatory disturbance created by the mutation. It also includes the downstream impact of the affected protein within the pathway encoded by that geneBiochemical State
Each phenotype develops a distinct physiological profile shaped by its pathway dynamics.Trait Expression
This physiological state produces both the autism traits and the comorbid traits associated with that phenotype.Individualized Expression
Each individual also encounters environmental experiences that are unique to them as an individual, and thus each individual will also have their own unique profile within their phenotype cluster.
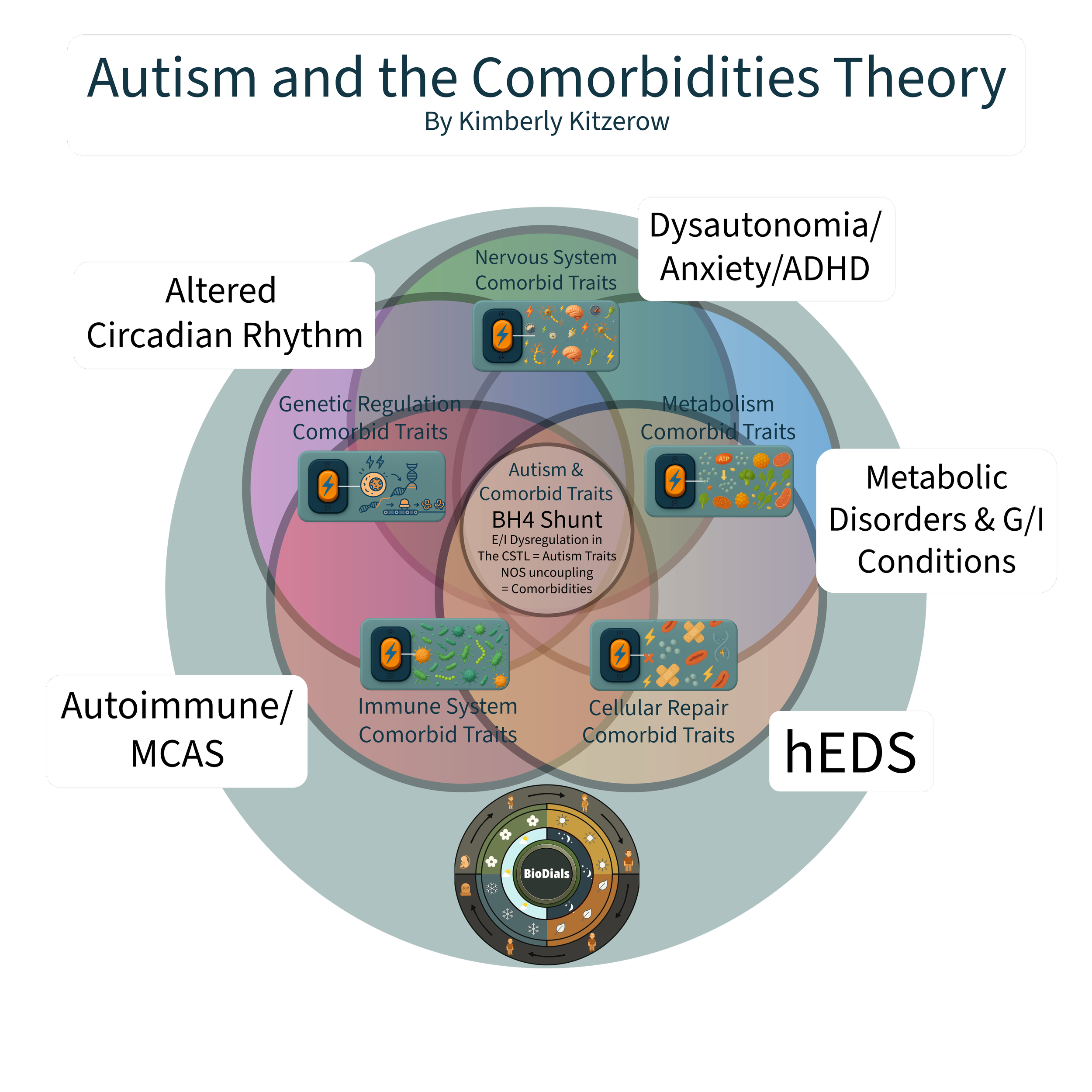
Autism and the Comorbidities Theory
Autism phenotypes can be defined by the allostatic biochemical state produced by their gene mutations. This state explains why autism traits and comorbidities consistently cluster together.Development Into Neurodivergent Biochemistry
BioToggles do not need to be genetically locked to influence traits, they can also be situationally flipped, or chronically stuck. This understanding initiated the development of Neurodivergent Biochemistry. Neurodivergent Biochemistry examines how the body’s survival systems, genetics, and environment work together to shape development, health, and learning. It focuses on how regulatory system activation influences physiological function and developmental outcomes over time.
This sequence is the complete mechanistic logic of my framework.
-
To make the theory teachable and practically useful, I created educational and analytical frameworks for visual and explanatory purposes::
NeuroToggle — neuroplasticity strategies for building, expanding, strengthening, and timing neural connections (used in my daughter’s speech progress).
BioToggles — the five regulatory systems (immune, metabolic, cellular repair, nervous system, genetic regulation).
BioDials — time-based protein synthesis cycles (circadian, circannual, and developmental).
Neurodivergent Biochemistry — a systems-level integration explaining upstream and downstream impacts of allostasis in various contexts considering the variables of the BioToggles becoming situationally flipped, chronically stuck, or genetically locked and how that impacts the timer regulated flow-state of protein synthesis within the BioDials.
Together, these frameworks make the Autism and the Comorbidities Theory and Neurodivergent Biochemistry concepts accessible
-
My work was documented through timestamped publications, presentations, and my book:
Papers and presentations posted on ResearchGate
Materials hosted on my websites - currently kimberlyedu.org (previously kimberlyseduationalresources.org and kimberly102347.com. I changed platforms from wordpress to squarespace over the last several years.)
Videos and visual explanations distributed online to wide audiences (instagram and tiktok)
My book, Discovering Autism and the Comorbidities, published on Amazon in September 2023
As an autistic individual and educator with degrees in education and special education with a minor in instruction strategies graduating summa cum laude from UW Superior, I often express complex ideas most effectively through visual formats such as infographics and recorded presentations. These formats are nonetheless publicly available, timestamped, and constitute intellectual property. They establish priority of authorship and fall under the protections of research integrity standards.
Concerning Overlaps
Following publication and public dissemination of my theory and frameworks, I began to see political and academic overlaps with my work as well as emerging research that, while concerning, indicates that my theory does have validity. If a theory is precise, it will have explanatory power and all past, present, and future research will converge around it.
Overview
Brazil Is claiming credit for the biochemical mechanism I uncovered: the BH4 Shunt that causes autism traits and comorbid traits to cluster.
I petitioned Stanford’s Neurodiversity Project in January of 2024 for help, and they recently published a paper utilizing the downstream mechanism of upregulated transamination pathways from the BH4 Shunt dysregulating E/I balance in a piece of the CSTL to create an effective “cure” for autism’s classic core symptoms. Again, uncited.
Princeton researchers tested my initial hypothesis, the Exclusivity Principle, directly. They took the core prediction of my framework that autism traits and comorbidities are inseparable because they arise from shared gene-pathway mechanisms, and tested it without citation while presenting it as their own hypothesis.
Northeastern University, through a Harvard affiliated study, recognized the BioToggles framework (in structure) and delineated the anatomical framework of the Nervous System BioToggle.
In 2024 people pushed me to “prove” my hypothesis with statistics. I refused because forcing a functional systems model into statistics breaks the logic and distorts the biology.
Statistics can only describe patterns in existing data, not explain the biology behind them. Princeton dismissed that, used statistics anyway, and ended up distorting the pattern.
Princeton and Autism and Comorbid Trait Clustering
-
The Princeton team tested my initial hypothesis, the Exclusivity Principle, directly. They took the core prediction of my framework that autism traits and comorbidities are inseparable because they arise from shared gene-pathway mechanisms, and tested it without citation while presenting it as their own hypothesis. I believe this may have been a falsification attempt, and when they realized the data supported this theory instead of falsifying it they published in their own names. They put out a press release saying that in 2025 they are responsible for a paradigm shift that I created in 2023.
-
They tested my Exclusivity Principle by asking:
• Do autism traits and comorbidities cluster together in stable, phenotype-specific patterns?
• Do those trait clusters correspond to distinct biochemical pathway signatures?These are the direct tests of my hypothesis, not independently derived conclusion from their own study.
Also, parallel discovery is not possible two years post public dissemination.
-
They had to have come up with the hypothesis somehow, and there is typically variation when independently derived. Theirs was identical to mine.
It is, however, possible they asked AI for testable ideas.
Although, regardless, it is the responsibility of the researchers conducting research to make sure they educate themselves on all prior literature on the topic. Primary sources must be cited. If AI gave them the source they would have had to have find the source. AI does not generate ideas on its own. If it is AI generated, it comes from somewhere. It doesn’t “think” it regurgitates what is already known.
I would have come up on a keyword search. My work is on ResearchGate and DOI indexed. Even if it wasn’t, if they want to test an existing hypothesis, they have to cite the source. Not doing so is plagiarism.
-
I developed the exclusivity principle hypothesis in 2023, and spent 2024 refining the conclusions. To test my hypothesis, I created a biochemical network and compared autism biomarkers to find points of dysregulation. The Autism and the Comorbiesis Theory is my conclusion, not the hypothesis.
They ran their initial GitHub first commit May 24, 2024. This is the mark of the beginning of the project. Their preprint states their ShinyGO search was ran June 3rd, 2024. They submitted their final paper to Nature July 25th, 2024. That is two months from start to finish.
-
In reviewing the Princeton preprint and subsequent Nature publication, there are multiple points of concern:
Hypothesis: In their preprint, it states a hypothesis identical to the Exclusivity Principle: It is implausible for autism traits and comorbid traits to co-occur without a joint root mechanism in each phenotype, arising from the same biochemical mechanism. This mechanistic hypothesis was removed. In the final Nature version, this mechanistic framing was removed, leaving only statistical correlations, although the framework’s structure was retained.
Methodological sequence: I broke down autism and comorbid symptoms into physiological traits, mapped them to protein function, and clustered them via proteins regulating multiple biochemical pathways and the regulatory systems influencing them, thereby generating mechanistic phenotypes. The Princeton version followed the same process but substituted questionnaire-derived traits and statistical clustering, ultimately linking clusters to pathways through correlation-based enrichment.
Post hoc reasoning: A peer reviewer explicitly noted reliance on post hoc reasoning and the absence of functional explanatory logic, underscoring the lack of mechanistic grounding.
Jigsaw Puzzle Methodology: A co-author publicly described their work using the term “jigsaw puzzle,” The Autism and the Comorbidities Theory was created using the the Jigsaw Puzzle Research Methodology, as described in a prior publication. This is due to the nature of piecing together a biochemical network out of raw protein data from Uniprot, and comparing autism biomarkers to find points of dysregulation liek a puzzle. The Princeton study similarly decomposes traits, clusters them, and retrospectively aligns them with biochemical pathways, but does so without acknowledgment of that methodology or its mechanistic structure.
-
Exhibit A: Framework comparison showing parallel structures in exclusivity, traits, clusters, proteins, and explanations.
Exhibit B: Preprint versus Nature excerpts showing explicit mention of my hypothesis in the preprint and its removal in the published version.
Exhibit C: Exclusivity principle comparison showing their rephrasing of my functional exclusivity theory as statistical exclusivity.
Exhibit D: Methodological mirroring contrasting my mechanistic workflow with their statistical substitution.
Exhibit E: Timeline showing my prior dissemination, Princeton’s ShinyGO results dated June 3, 2024, and their submission to Nature on July 25, 2024.
Exhibit F: Published book excerpt (September 2023, Amazon) articulating the exclusivity principle prior to Princeton’s work.
Exhibit G: Visual documentation of my contribution to the exclusivity principle, shared publicly through timestamped videos and infographics, demonstrating my original framing of autism and comorbidity clusters.
Preprint: https://www.medrxiv.org/content/10.1101/2024.08.15.24312078v1
Nature Study: https://www.nature.com/articles/s41588-025-02224-z
Princeton
Princeton recently released a study categorically delineating autism into biological phenotypes based on how the gene mutations within each cluster impact biochemical pathway activity uniquely in each phenotype.
What Princeton Tested
The Princeton team tested my initial hypothesis, the Exclusivity Principle, directly. They took the core prediction of my framework that autism traits and comorbidities are inseparable because they arise from shared gene-pathway mechanisms, and tested it without citation while presenting it as their own hypothesis. I believe this may have been a falsification attempt, and when they realized the data supported this theory instead of falsifying it they published in their own names. They put out a press release saying that in 2025 they are responsible for a paradigm shift that I created in 2023.
They tested my Exclusivity Principle by asking:
• Do autism traits and comorbidities cluster together in stable, phenotype-specific patterns?
• Do those trait clusters correspond to distinct biochemical pathway signatures?
These are the direct tests of my hypothesis. I do not believe they are not independent insights. They are too closely aligned with testing my exclusivity principle to be original. Parallel discovery also is not possible two years post public dissemination, especially a viral and globally known one.
Starting from that assumption, they used the SPARK dataset to statistically sort children into groups based only on which traits and health issues tended to show up together. SPARK is a very large autism database that includes both genetics and detailed questionnaire data for thousands of autistic children and their siblings.
Then, they figured out which gene mutations each person had in each comorbidity cluster "bucket" from the SPARK study data, and ran a bioinformatics tool called ShinyGo to figure out which biochemical pathways were involved with those genes.
The ShinyGo analysis was ran on June 3, and the full manuscript was submitted to Nature by July 25.
They then went one step further and asked when and where in brain development those genes tend to be active.
In my framework this is the impact of the gene mutations on the BioDials framework. I've had this mechanism outlined in my papers, and just recently created a visual and terminology for this.
The BioDials, as I define them, are the timing cycles that maintain protein synthesis over time that maintain physiological function and development/repair over time.
My approach begins with the mechanism. I mapped how the system itself functions. When the mechanism is understood first, the pattern is reliable because it comes from biology, not from the people being measured. Once the mechanism is mapped, individuals can identify where they fall within the system. This becomes a diagnostic blueprint rather than a statistical guess.
Statistical categories shift when the population shifts. Mechanistic categories do not. Genes are 99.9% conserved across all humans. This is why statistics cannot uncover or replace the functional mechanism that produces the autism and comorbidity pattern.
This is why a peer reviewer recognized they were using post hoc logic, and made note of it. It is illogical to cluster based on human characteristics first, then go looking for the biology behind why the traits cluster, and call it a biologically delineated phenotype system.
Areas of Concern
In reviewing the Princeton preprint and subsequent Nature publication, I observed multiple points of overlap with my work that raise the need for research integrity review:
Hypothesis: In their preprint, the authors stated they tested the same hypothesis I had already published, that autism and comorbidities arise from distinct biochemical pathway dysfunctions. In the final Nature version, this mechanistic framing was removed, leaving only statistical correlations, although the framework’s structure was retained.
Exclusivity principle: Their paper incorporates my principle that autism and comorbidities cannot occur simultaneously without a shared functional mechanism. They rephrased this without citation as: “As traits are not independent, they cannot be separately associated with patterns of genetic variation.”
Methodological sequence: I broke down autism and comorbid symptoms into physiological traits, mapped them to protein function, and clustered them via proteins regulating multiple biochemical pathways and the regulatory systems influencing them, thereby generating mechanistic phenotypes. The Princeton version followed the same process but substituted questionnaire-derived traits and statistical clustering, ultimately linking clusters to pathways through correlation-based enrichment.
Post hoc reasoning: A peer reviewer of the Nature publication explicitly commented on the authors’ post hoc reasoning, observing that they failed to provide functional explanatory logic for their findings. This criticism highlights the lack of mechanistic grounding in their framework compared to mine.
Jigsaw Puzzle Methodology: A co-author publicly described their work using the term “jigsaw puzzle,” which is a phrase unique to me and my work. I originated the Jigsaw Puzzle Research Methodology that I used to delineate my Autism and the Comorbidities Theory, which synthesizes research by constructing a biochemical network of human gene-coded proteins organized into functional sequences. Biomarkers are then overlaid to identify points where biochemical flow is disrupted, producing predictable downstream pathological consequences. These disruptions can be assessed individually or clustered by proteins that regulate multiple pathways, thereby explaining conditions with trait checklists such as autism and its comorbidities. Princeton’s study employs a statistical clustering approach that parallels this methodology, breaking down autism traits and comorbidities into clusters and then applying post hoc biochemical pathway logic. This overlaps conceptually with my framework but lacks the mechanistic foundation that is central to my work.
Taken together, these parallels suggest more than coincidental overlap and raise legitimate questions about the relationship between my prior work and Princeton’s publications. I have contacted their research and integrity office and they have escalated it to the Dean of Faculty for review.
Supporting Materials
Exhibit A: Framework comparison showing parallel structures in exclusivity, traits, clusters, proteins, and explanations.
Exhibit B: Preprint versus Nature excerpts showing explicit mention of my hypothesis in the preprint and its removal in the published version.
Exhibit C: Exclusivity principle comparison showing their rephrasing of my functional exclusivity theory as statistical exclusivity.
Exhibit D: Methodological mirroring contrasting my mechanistic workflow with their statistical substitution.
Exhibit E: Timeline showing my prior dissemination, Princeton’s ShinyGO results dated June 3, 2024, and their submission to Nature on July 25, 2024.
Exhibit F: Published book excerpt (September 2023, Amazon) articulating the exclusivity principle prior to Princeton’s work.
Exhibit G: Visual documentation of my contribution to the exclusivity principle, shared publicly through timestamped videos and infographics, demonstrating my original framing of autism and comorbidity clusters.
Preprint: https://www.medrxiv.org/content/10.1101/2024.08.15.24312078v1
Nature Study: https://www.nature.com/articles/s41588-025-02224-z
Princeton and Autism and Comorbid Trait Clustering
Princeton Tested My Central Hypothesis and Claimed the Concept was Theirs
Northeastern University and the BioToggles
Northeastern University, through a Harvard affiliated study, recognized the BioToggles framework (in structure) and delineated the anatomical framework of the Nervous System BioToggle.
Harvard’s 2025 Neuron paper introduces a new conceptual framing of allostasis that did not appear anywhere in their prior publications, grants, or models. Historically, their work consistently presented allostasis as one unified system, distributed across interoceptive and visceromotor networks. Their earlier papers (2016–2024) described a single integrated allostatic–interoceptive architecture that dynamically predicts and regulates energy needs.
However, in this new Neuron paper, they introduce entirely new language that aligns directly with the structural logic of my BioToggles framework. For the first time, they describe allostasis as being maintained by multiple interacting regulatory systems, and then immediately proceed to delineate the anatomical structure of the nervous system’s regulatory role. This mirrors my functional model of categorical regulatory systems (BioToggles) and the Nervous System BioToggle specifically.
What Existed Before My Work
Prior to the publication of my BioToggles framework on August 2, 2024, Harvard’s allostasis and interoception research consistently described:
one unified allostatic system
with the interoceptive network and visceromotor network treated as components of the same system, not separate regulatory systems
no framework dividing brain-driven regulation into multiple categorical regulatory subsystems
no language defining the nervous system as its own regulatory unit within a broader system of interacting regulatory systems
Their terminology consistently emphasized integration, not categorical differentiation.
What Is New in the 2025 Harvard Study
The 2025 Neuron paper departs from their previous theoretical model and introduces concepts that align directly with the structure of my BioToggles framework.
They write:
“In allostasis, many interacting regulatory systems collectively contribute to the survival of the organism.”
This is a structural shift.
It reframes allostasis as the product of multiple regulatory systems operating in coordination, which is the exact architecture defined in my BioToggles model.
This categorical, multi-system framing did not exist in any of their previous publications.
Areas of Concern
Conceptual Reframing: Harvard shifted from describing a unified allostatic system to describing many interacting regulatory systems.
This new terminology parallels the BioToggles framework and did not appear in any earlier Harvard publications.
Subsystem Delineation: Their anatomical mapping of the nervous system’s regulatory role reflects a new subsystem interpretation.
The detailed treatment of the nervous system as a distinct regulatory system is consistent with the Nervous System BioToggle and does not appear in their prior work.
Framework-Level Overlap: The multi-system structure introduced in the 2025 paper matches the defining architecture of the BioToggles model.
The framing of coordinated regulatory systems that jointly maintain survival mirrors my published structure for understanding autism and systemic comorbidities.
Lack of Citation: The new categorical framework appears only after the BioToggles model was published.
Despite clear structural parallels, the authors do not cite or acknowledge this shift, even though it represents a departure from their historical work.
Supporting Materials:
Exhibit A Harvard: New multi-system regulatory framing
Quote: “In allostasis, many interacting regulatory systems collectively contribute to the survival of the organism.”
Explanation: First appearance of plural regulatory systems in Harvard’s work. Establishes a categorical multi-system architecture aligned with the BioToggles model.
Exhibit B Harvard: Prior unified allostatic system (October 2024)
Quote: “These results reinforce the existing evidence for a whole-brain system that supports the modeling and regulation of the body’s internal milieu.”
Explanation: Reinforces their historical single-system framing before the introduction of plural regulatory systems in 2025.
Quote: “Large swaths of brain tissue are centrally involved in allostasis, with the rest serving as association regions for the predictive, efficient regulation of the body.”
Explanation: Separates core allostatic regions from distinct body-regulatory regions, creating categorical regulatory functions rather than a unified system. This represents an early move toward multiple regulatory systems.
What Is New in the 2025 Naviaux Update
The 2025 update to the Cell Danger Response model departs from Naviaux’s earlier structure and mimics concepts that align directly with the architecture of my Autism and the Comorbidities Theory.
For the first time in his work, he frames autism and its medical comorbidities together as a single mechanistically linked biological progression. He writes that autism is associated with “multimodal sensory and chemical over-responsivity and an increased risk of several chronic medical conditions that co-occur with ASD”. This unified framing of autism and comorbidities did not exist in his prior publications. It is the central premise of my Autism and the Comorbidities Theory, which establishes the BH4 shunt as the mechanistic bridge linking neurodevelopmental traits and systemic comorbidities.
He also introduces a new three hit model that mirrors the progression defined in my Immune System BioToggle. Naviaux writes that the “three hits necessary and sufficient to cause ASD are: 1) DNA predisposition, 2) early exposure to CDR activating triggers, and 3) recurrence or persistence of CDR activating triggers for at least three to six months”. This staged progression did not appear in any of his earlier CDR publications. It now reflects the sequence I published in 2023, where the one (or more) of the stress regulatory response systems (BioToggles) becomes situationally flipped, then chronically stuck when gene mutations prevent resolution after an initial flip.
He introduces mechanistic elements that were not present in earlier versions of the CDR model, including the use of PKU to illustrate metabolic disruption, the adoption of the excitatory to inhibitory imbalance as a central outcome of prolonged activation, and the claim that stress driven signaling shifts the entire trajectory of child development, which mirrors my BioDIals framework. He states that this signaling “changes mitochondrial function, shifts the trajectory of child development, and produces the core symptoms of ASD”. These mechanisms align with work I previously published.
Areas of Concern
Conceptual Reframing: Naviaux has shifted from describing CDR as a unified metabolic response to describing autism and its comorbidities as a staged biological progression. This new structure reflects the BioToggle sequence of situationally flipped, chronically stuck when resolution fails, and genetically locked. None of this appears in his earlier work.
Adoption of the Autism and Comorbidities Framework: He now presents autism and its comorbidities as a single mechanistically connected pathological framework. His statement that ASD is associated with “an increased risk of several chronic medical conditions that co-occur with ASD” adopts the exact framing, and verbiage, that my Autism and the Comorbidities Theory introduced. He had never previously offered a mechanism that links autism traits and comorbidities into a single framework.
Mechanistic Adoption: The updated paper introduces BH4 dependent stress regulation of the excitatory to inhibitory balance. Naviaux states that prolonged activation “prevents the normal excitatory to inhibitory reversal in extracellular ATP and GABA signaling networks” and then links this failure to mitochondrial dysfunction and activation of microglia and astroglia. This mechanism is a core component of my BH4 Shunt model.
Use of PKU While Avoiding BH4: The update uses PKU as the model’s key developmental analogy and states that “the three hit model described above for ASD also applies to the current paradigm for the diagnosis and treatment of phenylketonuria (PKU)”. Earlier in the paper he explains that the enzyme responsible for phenylalanine metabolism requires the cofactor tetrahydrobiopterin. He writes that PAH converts phenylalanine to tyrosine using “iron, molecular oxygen, and the cofactor tetrahydrobiopterin”.
PKU is a BH4 dependent disorder. Using PKU to illustrate early biochemical disruption while avoiding any mention of BH4 within the autism mechanism is scientifically inconsistent unless the upstream mechanism is being intentionally avoided. The BH4 Shunt is the central biochemical mechanism I am known for, and PKU is one of its clearest clinical demonstrations.
Excitatory to Inhibitory Imbalance Integration: The updated paper positions failure of the excitatory to inhibitory reversal as a core downstream consequence of prolonged CDR activation. The relevant quote is that prolonged activation “prevents the normal excitatory to inhibitory reversal in extracellular ATP and GABA signaling networks”. He does not specify what this mechanism is. In my framework the BH4 Shunt upregulates transamination pathways that dysregulate this balance through unregulated glutamate activity and synthesis, dysregulatin the cortico-striatal-thalamic-loop that is responsible for movement, habit formation, and reward, thus resulting in core autism traits. This is one of the defining downstream effects of BH4 dysregulation in my published work. This mechanistic connection does not appear in earlier CDR publications.
Lack of Citation: All of these elements appear only after my frameworks were published. The structural alignment with the BioToggles, the unification of autism and comorbidities, the staged progression, the stress driven E/I imbalance, and the selective use of PKU without BH4 emphasis are introduced for the first time here without acknowledgment of the original source.
Supporting Materials
Exhibit A Naviaux Introduction of Three Hit Structure
Quote: “The three hits necessary and sufficient to cause ASD are: DNA predisposition, early exposure to CDR activating triggers, and recurrence or persistence of CDR activating triggers for at least three to six months.”
Explanation: This is the first appearance of a staged progression in CDR theory. It mirrors the stress regulated BioToggle sequence.
Exhibit B Naviaux Earlier Unified CDR Model
Quote: “The Cell Danger Response is a unified metabolic response conserved across evolution.”
Explanation: Earlier CDR work describes a single mechanism with no staged progression and no unified autism and comorbidity framework.
Exhibit C Naviaux Use of PKU Without BH4
Quote: “The protein made by the PAH gene is an enzyme that converts phenylalanine to tyrosine using iron, molecular oxygen, and the cofactor tetrahydrobiopterin.”
Explanation: He names BH4 in the context of PKU but omits BH4 entirely when applying the same developmental mechanism to autism, even though he states that PKU exemplifies his three hit model.
Exhibit D Naviaux Excitatory to Inhibitory Imbalance
Quote: “Prolonged CDR activation prevents the normal excitatory to inhibitory reversal in extracellular ATP and GABA signaling networks.”
Explanation: He adopts the E to I imbalance as a core outcome, which matches the downstream mechanism of the BH4 Shunt and the Immune System BioToggle.
Naviaux, the 3-Hit Model, and my BioToggles Framework
Naviaux rebuilt his updated cell danger response framework by adopting the full structure of my autism and the comorbidities model through the stress BioToggles response which leads into the BH4 shunt mechanism which produces the excitatory to inhibitory imbalance in circuitry that generates autism traits and the developmental progression BioDial logic, as well as simultaneous stress induced comorbidities, incorporating these mechanisms into his three hit model without citing the original source.
Brazil and the BH4 Shunt
In February 2025, researchers at the Universidade Federal de Santa Catarina published a paper titled “Tetrahydrobiopterin and Autism Spectrum Disorder: A Systematic Review of a Promising Therapeutic Pathway” (Brain Sci. 2025, 15(2), 151). This review presents central components of a framework I had already published and documented.
My work was established in September 2023 with the release of my book Discovering Autism and the Comorbidities Along the BH4 Pathway (Amazon) and expanded through multiple ResearchGate publications, including:
Autism and the Comorbidities Along the BH4 Pathway (Jan 2024, DOI: 10.13140/RG.2.2.23124.37761/1)
The BH4 Pathway as an Allostatic Mechanism in the Pathology of Autism and Systemic Comorbidities (Aug 2024, DOI: 10.13140/RG.2.2.19905.98401/2)
This body of work established the BH4 pathway as a central pathological mechanism that biochemically links autism and its comorbidities. My contributions were timestamped and publicly available well before the submission of the Brazilian review.
I contacted the journal that published this paper, MDPI stated that Alexandra Latini is claiming this hypothesis is a novel conclusion to the systematic review done in this paper. This raises several concerns:
This Brazilian researcher, Alexandra Latini, published a paper on BH4 and its role in the entire human body just a few months before I published mine. She covered autism in it, in a very limited section under the tile "Non-BH4-Linked Genetic Deficiencies of BH4 Metabolism" it is positioned next to rabies and malaria. She has never written a paper about autism, and has never used the word comorbidities in any prior paper (to my knowledge). This paper outlines BH4 and its central role in our stress response system as a whole.
MDPI stated that she couldn't have taken my hypothesis because she used research from 1967-2021 in her review. Then, they cited her 2023 paper as evidence that she has mentioned BH4 and autism together before. However, if her novel hypothesis was autism and the comorbidities were pathologically linked along the BH4 pathway, the autism section would have been a focus of the paper and not situated next to malaria under the heading "non-BH4-Linked Genetic Deficiencies of BH4 Metabolism." Latini also cites research through 2024 in this paper. The timeline doesn't add up. It appears that she may be trying to backdate this new paper off of an old keyword search she did for her 2023 paper after coming across my work, or Al introduced herto it.
BH4 has been known to be linked to autism, but a causal framework pathologically linking autism and the comorbidities along the BH4 pathway had not been formalized until my model. Before my model, they had known BH4 biomarkers were low/high in autistics, but they did not know why or investigate the implications pathologically as far as mechanistic logic goes. There is a big difference between "hey I noticed these biomarkers were low and I wonder what happens if we treat it" vs. "hey these levels dysregulated, as well as the pathways that impacts, lets build a pathological framework behind why that can manifest into autism and comorbid trait clusters."
the paper states the intent of the paper is to investigate the role of this metabolic pathway in the pathophysiology of autism, they conducted a systematic review of existing biomarkers associated with the BH4 pathway in the biological fluids of individuals with autism. No new data was collected. Both things can't be true at the same time. If that was a novel conclusion, it would be in the discussion and not the intro. This hypothesis of autism and the comorbidities being pathologically linked along the BH4 pathway has never been discussed in any prior paper. I created it and was called a pseudo-scientist for years for it. Which is why it was so jarring.
Areas of Concern
Use of my hypothesis as prior knowledge without citation
In their introduction, the authors listed my hypothesis as though it were already established knowledge, without mentioning where it came from. The BH4 Shunt mechanism and its role in linking autism to comorbidities is my original contribution, first defined and published by me.Systematic review anchored to my theory
The review frames its research question directly on the BH4 pathway as a causal mechanism for autism comorbidities. This mirrors my published hypothesis but provides no citation. BH4 has been mentioned in autism research for decades, but my work delineated the biochemical mechanism that connects autism to its comorbidities through the BH4 pathway.Methodological and registration issues
The methods section acknowledges the review was not registered during the planning stage, which increases the risk of duplication. Their search strategy was limited to BH4, nitric oxide, and ASD, excluding other BH4-dependent pathways required to construct the complete Autism and the Comorbidities framework. Despite this, the paper presents the completed theory as though it had been their intent all along. There is also no public record of the work before their December 31, 2024 submission to MDPI, and it relied on a universal grant with no detailed proposal.